Fasting has long-held associations with mental and bodily repair, from religion asceticism to detox diets. A new study points to one reason fasting may be good for our bodies, with evidence that a 24-hour gap in eating can flick a metabolic switch for regeneration in our digestive system.
By studying the effects of fasting on mice, a team of researchers at MIT found that just one day of calorie restriction substantially improves the ability for stem cells in the intestine to regenerate. Not only does this underline the beneficial effects of short periods of fasting, but it could also pave the way for drugs that stimulate the effects in cancer patients.
As we get older, our intestinal cells get worse at regenerating, meaning our bodies are less able to fight off infections and diseases in our gut. In the fasting mice, however, cells began to break down fatty acids instead of glucose. This stimulated the stem cells to become more regenerative.
“This study provided evidence that fasting induces a metabolic switch in the intestinal stem cells, from utilising carbohydrates to burning fat,” says David Sabatini, an MIT professor of biology and senior author of the paper, published in Cell Stem Cell.
“Interestingly, switching these cells to fatty acid oxidation enhanced their function significantly. Pharmacological targeting of this pathway may provide a therapeutic opportunity to improve tissue homeostasis in age-associated pathologies.”
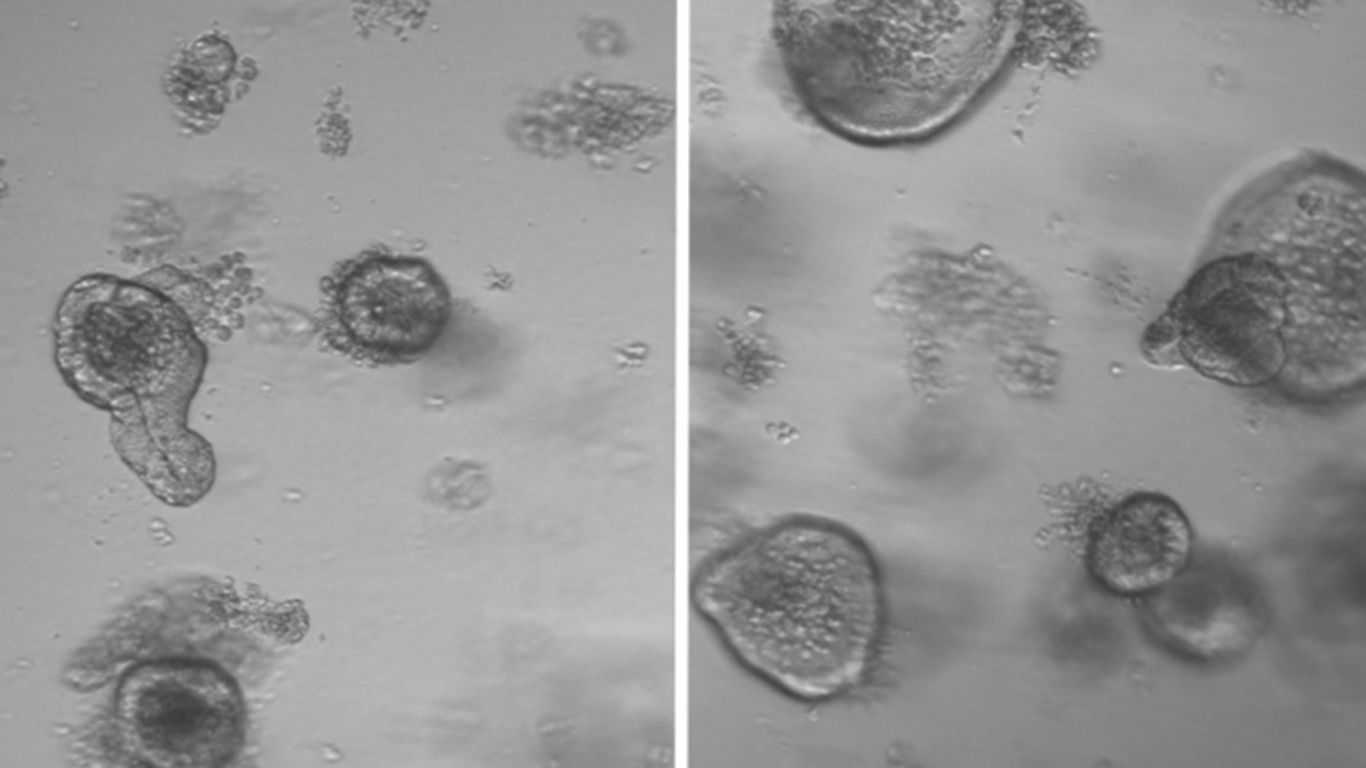
(Intestinal organoids from mice that fasted for 24 hours (R) and from mice that did not fast (L). Credit: Maria Mihaylova and Chia-Wei Cheng)
The scientists took samples of the mice’s intestinal cells after a period of 24-hour fasting, then grew them in the lab to determine their ability to produce an “organoid”, in this case a kind of mini-intestine. They found that the regenerative capacity of the mice that had fasted was double that of mice that hadn’t fasted.
“It was very obvious that fasting had this really immense effect on the ability of intestinal crypts to form more organoids, which is stem-cell-driven,” said lead author, Maria Mihaylova. “This was something that we saw in both the young mice and the aged mice, and we really wanted to understand the molecular mechanisms driving this.”
To glean the reasons for the increased regeneration, the researchers sequenced the messenger RNA of the stem cells, and found that fasting flips a “switch” by activating transcription factors called PPARs, which turned on genes involved with metabolising fatty acids. The result is the cell metabolises fatty acids instead of carbohydrates.
Crucially, the scientists found they could imitate this effect with a molecule that mimics the effects of the PPARs. This suggests that drug treatment could one day be developed to similarly flip the metabolic “switch”, and help regeneration in the intestine. This could have massive benefits for the elderly, and for cancer patients receiving chemotherapy, which tends to harm cells in the digestive system.
“In a beautiful set of experiments, the authors subvert the system by causing […] metabolic changes without fasting and see similar effects,” says Jared Rutter, a professor of biochemistry at the University of Utah School of Medicine, who wasn’t involved in the research.
“This work fits into a rapidly growing field that is demonstrating that nutrition and metabolism [have] profound effects on the behavior of cells and this can predispose for human disease.”
Disclaimer: Some pages on this site may include an affiliate link. This does not effect our editorial in any way.


